T-cell therapy of MYD88L265P-dependent B-cell lymphoma
Keywords
TCR,T-cell therapy,oncology,B-cell lymphoma,MyD88,neoantigen
Challenge
Diffuse large B-cell lymphomas (DLBCL) exhibit high degree of genetic heterogeneity. MyD88L265P is a prominent genetic aberration responsible for fostering tumor growth in this heterogenous lymphoma entity. In particular 17% of DLBCL and 42% in primary central nervous system lymphoma (PCNSL) patients carry the oncogenic mutation. The resulting aberrant scaffold protein would theoretically provide a suitable target for therapeutic intervention but is almost not addressable by conventional pharmacological means.
Technology
Novel human TCRs for targeting the mutant epitope of MyD88L265P have been developed for an adoptive TCR cell therapy approach in HLA-B7 positive patients. This cell therapy approach offers a novel therapeutic option for patients with relapsed and/or refractory DLBCL and primary CNS lymphoma in a personalized fashion (stratification by presence of somatic mutation). TCR redirected T-cells are able to selectively recognize and kill only those target cells presenting the MyD88L265P mutant epitope in an HLA-B7 restricted manner. This technology provides an opportunity for a straightforward development path towards clinical application in a defined patient population.
Commercial Opportunity
This opportunity is available for in-licensing or (pre)clinical (co-)development towards First-in-Human with retrovirally or non-virally modified TCR-T-cells.
Development Status
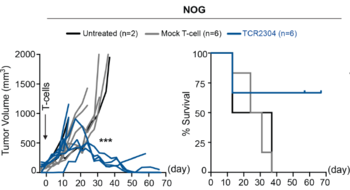
Extensive analysis regarding specificity, reactivity, cytotoxicity and safety for various TCRs has been carried out in vitro in multiple Non-Hodgkin lymphoma cells as well as in suitable in vivo experiments. A lead TCR has been identified.
A single-arm, multicenter phase I study in patients with r/r MyD88L265P-mutated DLBCL including PCNSL is planned (First Patient In Q3 2024). Patients will receive increasing doses of retrovirally transduced TCR-T-cells. The primary endpoints of this first-in-human study are safety and determination of the maximum tolerable dose of a single infusion of TCR-T-cells.
Patent Situation
Patent applications pending in CA, CN, EP, US (based on WO2020/152161, priority January 2019).
Further Reading
Çinar, Ö., et al., «High-affinity T-cell receptor specific for MyD88 L265P mutation for adoptive T-cell therapy of B-cell malignancies», J Immunother Cancer. 2021 Jul;9(7):e002410. doi: 10.1136/jitc-2021-002410.
Weber, A.N.R., et al., «Oncogenic MYD88 mutations in lymphoma: novel insights and therapeutic possibilities». Cancer Immunol Immunother. 2018 Nov;67(11):1797-1807.